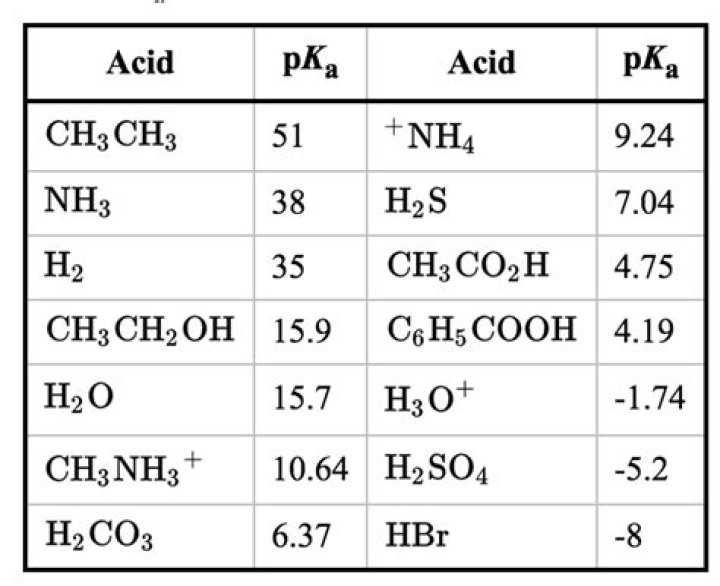
Answer and Explanation: The pKa of CH3NH3Cl C H 3 N H 3 C l is 10.65.
Is CH3NH3 a strong base?
CH3NH3+ acts as a acid because Brønsted-Lowry acids give away a proton. It is the exact opposite of acid in chemistry. Methylamine is a weak base.
Is CH3NH3+ A Lewis base?
In the above reaction, methylamine accepts a proton from water and is thereby a Bronsted base. For the reverse reaction, the opposite is true, as CH3NH3+ donates a proton to hydroxide, classifying it as a Bronsted acid. Therefore, CH3NH3+ is the conjugate of methylamine. The quick answer is that (CH3)2NH is a base.
What is the pKa for CH3NH2?
I think you must be asking name of CH3NH2 which is methyl amine . Instead CH3NH3+ is just conjugate acid of methyl amine. And name of CH3NH3+ must be methyl ammonium ion . As a base, it’s 15.9 or so (that is, the pKa of H2O).
What is the pKa of H3O+?
In water, pKa(H3O+) is └1.7 and pKa(NεHis) is about 7.
What is the pKa of CH3 3COH?
The pKa of tert-butanol ((CH3)3COH; conjugate acid of (b)) is 17.
Is nh4no2 acidic basic or neutral?
2 Answers By Expert Tutors
So yes, it is a weak acid (NH4+) and weak base (NO2-).
Why is CH3NH2 a Lewis base?
So, HCl accepts the lone pair of the electron, therefore, it is Lewis acid and CH3NH2 donates the lone pair of the electron, therefore, it is Lewis base.
Why is CH3NH2 a base?
Methylamine is a Bronsted base, as it can accept a proton from water.
Is ClO an acid or base?
Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−.
Is CH3NH2 a primary amine?
It is a primary aliphatic amine, a one-carbon compound and a member of methylamines.
What type of compound is CH3NH2?
Methylamine is an organic compound with a formula of CH3NH2. This colorless gas is a derivative of ammonia, but with one hydrogen atom being replaced by a methyl group. It is the simplest primary amine.
What is the functional group of CH3NH2?
The name of the compound CH3NH2 is Methlyamine The functional group present in it is Amine that has a suffix-NH2.
What is the pH of H3O+?
pH = – log [H3O+]. The hydronium ion concentration can be found from the pH by the reverse of the mathematical operation employed to find the pH. [H3O+] = 10-pH or [H3O+] = antilog (- pH) Example: What is the hydronium ion concentration in a solution that has a pH of 8.34?
What is pKa of HF?
It has multiple industrial and household uses (Box 15-1). The damage caused by HF is concentration dependent and largely related to the fluoride component rather than its corrosive hydrogen ion. Because HF is a relatively weak acid (pKa 3.8), it is largely nonionized and can penetrate cell membranes easily.